|
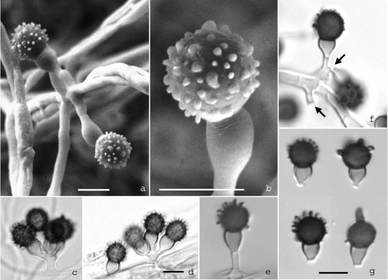
by Cliff Bueno de Mesquita I have finished analyzing data from an experiment I did looking at how adding plant litter (dead leaves) affects microbial communities in unvegetated soils. Since microbial growth in unvegetated soils can be limited by carbon, and plant leaves contain lots of carbon, we expected to see changes in microbial communities as different microbes increase in growth to decompose the leaves. Besides that, leaves also harbor their own microbial communities, which also get added to soil when they fall. Botrytis caroliniana (Figure 1) is a species of fungus that was abundant in plant litter but also varied among plant species. B. caroliniana was especially abundant in the leaves of the mountain sorrel plant Oxyria digyna and less abundant in the grass Deschampsia cespitosa and the cushion plant Silene acaulis. Botrytis is a relatively well studied fungal genus with 22 described species, which typically cause disease in plants (Williamson et al. 2007). Botrytis species can be both biotrophs (eating living material) or necrotrophs (eating dead tissues). Several books have been written about the genus, focusing on their negative effects on important crop species (Elad et al. 2007). For example, the species Botrytis cinerea is reported to cause gray mold disease on 200 crop species, including cabbage, lettuce, broccoli, beans, grapes, strawberries, raspberries, and blackberries (Figure 2, Williamson et al. 2007). Botrytis caroliniana gets its name because it was first isolated from blackberry in South Carolina (Li et al. 2012). It has also been reported to cause gray mold disease on strawberries in North Carolina (Fernández-Ortuño et al. 2012). It is closely related to Botrytis fabiopsis and Botrytis galanthina, which cause gray mold disease in broad bean and snowdrop plants (Li et al. 2012). Clearly, fungi in this genus are pathogens that can have major impacts on wild plants and agricultural crops. The market size for anti-Botrytis products was estimated at $15-25 billion USD (Elad et al. 2007). Now that we know that Botrytis caroliniana is abundant on Oxyria digyna leaves, we can conduct more specific research on the role of this fungus in plant health and plant communities in the alpine.
References: Elad Y, Williamson B, Tudzynski P, Delen N (eds) (2007) Botrytis: Biology, Pathology and Control. Springer, Dordrecht, The Netherlands Fernández-Ortuño D, Li XP, Wang F, Schnabel G (2012) First Report of Gray Mold of Strawberry Caused by Botrytis caroliniana in North Carolina. Plant Dis. doi: 10.1094/pdis-12-11-1018-pdn Li X, Kerrigan J, Chai W, Schnabel G (2012) Botrytis caroliniana , a new species isolated from blackberry in South Carolina. Mycologia 103:650–658. doi: 10.3852/11-218 Williamson B, Tudzynski B, Tudzynski P, Van Kan JAL (2007) Botrytis cinerea: The cause of grey mould disease. Mol Plant Pathol 8:561–580. doi: 10.1111/j.1364-3703.2007.00417.x
0 Comments
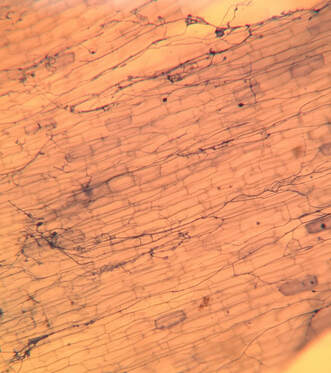
by Cliff Bueno de Mesquita We have started another round of microscopy here in the AMO. We are collaborating with the Suding Lab on understanding how nitrogen deposition and plant species interactions affect microbial communities. One aspect of the work, as in some of our other projects, is to quantify how much of the plant roots contain certain fungi. I have previously written about both arbuscular mycorrhizal fungi and dark septate endophytes, which are two groups of fungi that inhabit plant roots, but there is another – fine root endophytes – that I will tell you about today. Fine root endophytes (FRE) are a newly classified group of fungi that are characterized by having very fine hyphae. FRE were once considered to be arbuscular mycorrhizal fungi until further genetic work was able to distinguish them. Morphologically, they can be distinguished by their very fine hyphae, which are less the 2 µm wide – that’s 0.002 millimeters. But with the help of the microscope we can view them and identify them as FRE. They look like little fine blue lines (because we stain them with a blue dye) running all throughout the plant roots (Figure 1). Several studies have suggested that FRE can help plants uptake phosphorus, which means they may be functionally similar to arbuscular mycorrhizal fungi. FRE were once thought to be the species Glomus tenue, now reclassified as Planticonsortium tenue, but now several different morphologies have been observed and it is likely that there are several different species of fine root endophytes.
FRE are distributed globally, have been reported in 53 plant families, and have been found in all major habitat types. As we and others continue to quantify the amount of FRE in various plant species and various environments, we can learn more about the ecology of this potentially important fungal group. Additionally, more experiments are needed to learn more about the functioning of FRE. References: Orchard S, Hilton S, Bending GD, et al (2017a) Fine endophytes (Glomus tenue) are related to Mucoromycotina, not Glomeromycota. New Phytol 213:481–486. doi: 10.1111/nph.14268 Orchard S, Standish RJ, Dickie IA, et al (2017b) Fine root endophytes under scrutiny: a review of the literature on arbuscule-producing fungi recently suggested to belong to the Mucoromycotina. Mycorrhiza 27:619–638. doi: 10.1007/s00572-017-0782-z Walker C, Gollotte A, Redecker D (2018) A new genus, Planticonsortium (Mucoromycotina), and new combination (P. tenue), for the fine root endophyte, Glomus tenue (basionym Rhizophagus tenuis). Mycorrhiza 28:213–219. doi: 10.1007/s00572-017-0815-7 By Eli Gendron Diatoms are a unique life form that can be found in a wide range of environments, from the soils of the arctic and the some of the highest mountains in the world to freshwater lakes across the globe and throughout our oceans (Mann & Droop 1996). One of the characteristics that make diatoms so unique is the glass like shell they build out of silica which often leads them to have regular geometric shaped cells (see Figure 1). These single celled organisms often form colonies that take many forms such as the chains seen in Figure 1. Diatoms are almost always photosynthetic but several strains have been found to be capable of mixotrophy and heterotrophy, meaning they are capable of living off organic matter (Villanova et al. 2017; Tan & Johns 1996).  Figure 1. Image of Fragilaria capucina as seen under a microscope. Left: Image of Fragilaria capucina taken in Chico, Washington, USA; Chico Creek, 400x magnification, DIC – 13 Jul 2008. Right: Image of Fragilaria capucina taken in Bremerton, Washington, USA; Kitsap Lake, 1000x magnification, DIC – 11 Oct 2008. Images curated by http://www.algaebase.org/search/species/detail/?species_id=31451&distro=y#distro Diatoms such as Fragilaria capucina are a common occurrence in freshwater habitats around the world; however, they tend be low in abundance in oligotrophic (low nutrient) ecosystems. When an ecosystem is exposed to more nutrients than naturally occur, usually through pollution such as nitrogen in fertilizer run off, it becomes eutrophic (abundant nutrients) leading to blooms of previously rare organisms. Fragilaria capucina is one such diatom that is highly resistant to pollution and can often be found thriving in eutrophic conditions (Rott et al. 1998). Armed with this type of knowledge we can monitor communities of easily obtainable species to use as indicators of ecosystem status including catching pollution early. References: Mann, D.G. & Droop, S.J.M. (1996) Biodiversity, biogeography and conservation of diatoms. Hydrobiologia 336: 19-32 Rott E, Duthie HC, Pipp E. 1998. Monitoring organic pollution and eutrophication in the Grand River, Ontario, by means of diatoms. Can J Fish Aquat Sci 55:1443–1453. Tan, C.K. and Johns, M.R., 1996. Screening of diatoms for heterotrophic eicosapentaenoic acid production. Journal of Applied Phycology, 8(1), pp.59-64. Villanova, V., Fortunato, A.E., Singh, D., Dal Bo, D., Conte, M., Obata, T., Jouhet, J., Fernie, A.R., Marechal, E., Falciatore, A. and Pagliardini, J., 2017. Investigating mixotrophic metabolism in the model diatom Phaeodactylum tricornutum. Phil. Trans. R. Soc. B, 372(1728), p.20160404 by Cliff Bueno de Mesquita Happy new year’s eve from all of us here at the AMO! I am writing this post from little Rhode Island (where I grew up), so I decided to write about a local microbe. So this month’s microbe is a genus called Phytophthera. Phytophthera is a type of microorganism called an oomycete, which is different from, say, a bacterium or a fungus. Oomycetes are often described as being quite similar to fungi, but they form their own unique lineage on the phylogenetic tree of life. They are filamentous, spore-producing organisms that often function as either saprotrophs (organisms that decompose organic matter), or plant pathogens, living on plants and causing them harm. Unlike fungi, whose cell walls are made of chitin, Phytophthera cell walls are made of cellulose. There are 232 described species and species variations in the genus, though it is estimated that there could be as many as 200-300 more that we have not discovered yet! One of the most common tree diseases in Rhode Island is Phytophthera root rot, which occurs on beech trees. The Phytophthera oomycete in this case infests the beech tree roots, but infected trees will also show bleeding cankers on the trunks (Figure 1). Phytophthera root rot is a leading cause of death for older beech trees. But the Phytophthera genus is actually quite widespread and infects thousands of plant species, including important forest trees like chesnuts, oaks, and alders, and important crops such as strawberries, citrus trees, cucumbers, squash, soybeans, tomatoes and potatoes. Most notably, the species Phytophthera infestans caused the potato blight that led to the Great Irish Potato Famine of 1845-1849!
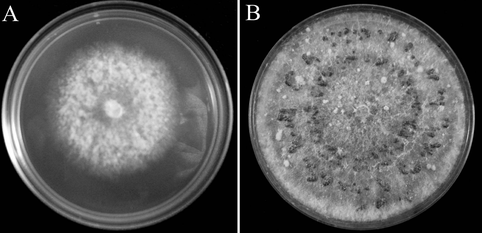
References: Goheen EM, Frankel SJ, eds. 2007. Phytophthoras in Forests and Natural Ecosystems. Albany, CA, USA: USDA Forest Service: General Technical Report PSW-GTR-221, 101–15. https://www.fs.fed.us/psw/publications/documents/psw_gtr221/psw_gtr221.pdf Kaiser Tree Preservation Co. 2018. Top 10 Tree Diseases in Rhode Island. http://kaisertree.com/services/plant-health-care-2-2/top-10-tree-diseases-in-rhode-island/ NCBI. 2018. Phytopthera. NCBI taxonomy. Bethesda, MD: National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Tree&id=4783&lvl=3&keep=1&srchmode=1&unlock Nowicki M, Foolad MR, Nowakowska M, Kozik EU. 2011. Potato and tomato late blight caused by Phytophthora infestans: An overview of pathology and resistance breeding. Plant Disease, 96: 4–17, doi:10.1094/PDIS-05-11-0458 by Cliff Bueno de Mesquita I hope everyone had a wonderful Thanksgiving and gave thanks to all of the fascinating microorganisms that we depend on. For this post I’ll write about a fungus that I was recently able to culture (Figure 1). The green culture seen in Figure 1 is characteristic of the fungal genus Trichoderma. This culture was isolated from the inside of a root of a grass that I have been working with for the Moving Uphill project. I harvested the grasses that I transplanted into unvegetated soil 2 years ago to see what I could culture and isolate for potential follow up studies. Trichoderma is a ubiquitous soil fungus but recent studies have shown that it can also colonize plant roots (Harman et al. 2004, Bae et al. 2011). Trichoderma is in the Hypocreaceae family, in the Ascomycota phylum. The taxonomy has been updated several times, but there are currently 89 species recognized in the genus (Samuels 2006). Trichoderma viride, perhaps the species I have in culture, was the first species described in the genus and is a green mold. Trichoderma aggressivum is another green mold that causes disease on cultivated button mushrooms (Samuels et al. 2002). In soil, Trichoderma is a saprotroph, eating organic carbon in the form of dead plant and fungal material. Trichoderma produces enzymes to break this matter down, and humans have used Trichoderma to produce industrial amounts of decomposition enzymes like cellulase and chitinase (Felse and Panda 1999). Cellulase breaks down cellulose, the main ingredient of plant cell walls, while chitinase breaks down chitin, the main ingredient of fungal cell walls.
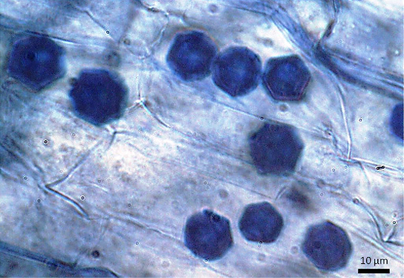
Inside of plant roots, the fungus produces a variety of compounds that can induce plant responses. Studies have shown that root colonization by Trichoderma frequently enhances root growth and development, plant productivity, resistance to abiotic stress, and nutrient uptake (Harman et al. 2004). Thus Trichoderma can be a very beneficial fungus for plants, but it has received much less attention than mycorrhizal fungi. It can be important for our crop plants too – Bae et al. (2011) found that Trichoderma helped delay disease in hot pepper plants. Perhaps it’s time for us to conduct some experiments with my cultures and see how they affect alpine plants! References: Bae, H; Roberts, DP; Lim, HS; Strem, MD; Park, SC; Ryu, CM; Melnick, RL; Bailey, BA (2011). Endophytic Trichoderma isolates from tropical environments delay disease onset and induce resistance against Phytophthora capsici in hot pepper using multiple mechanisms. Mol. Plant Microbe Interact. 24: 336–51. doi:10.1094/MPMI-09-10-0221. Felse, P.A, Panda, T. (1999). Taguchi Method. Enzyme and Microbial Technology. 40 (4): 801–805. doi:10.1016/j.enzmictec.2006.06.013. Harman, G.E., Howell, C.R., Viterbo, A., Chet, I., Lorito, M. (2004). Trichoderma species—opportunistic avirulent plant symbionts. Nature Reviews Microbiology. 2 (1): 43–56. doi:10.1038/nrmicro797. Samuels, G.J., Dodd, S.L., Gams, W., Castlebury, L.A., Petrini, O. (2002). Trichoderma species associated with the green mold epidemic of commercially grown Agaricus bisporus. Mycologia. 94 (1): 146–170. Samuels, G.J. (2006). Trichoderma: Systematics, the Sexual State, and Ecology. Phytopathology. 96 (2): 195–206. doi:10.1094/PHYTO-96-0195. by Cliff Bueno de Mesquita Happy Halloween! I just finished another round of root microscopy! We are interested in the ecology of fungal endophytes and often look at roots under the microscope to quantify how much of the root is colonized by fungi. While I focus on dark septate endophytes and mycorrhizal fungi (see previous posts), which are typically beneficial for plants, I also sometimes come across some pathogens in the roots. One such pathogen is the genus Olpidium. This genus of fungi has been of interest the agricultural field, as one species, Olpidium brassicae, has been shown to be a disease vector in a wide variety of plants including cucumber and lettuce plants. The fungus can infect lettuce roots, and transmit lettuce big-vein disease through its zoospores, which are the motile, asexual spores that Olpidium reproduces with (Tomlinson and Faithfull 1979). Another species, Olpidium radicale was shown to transmit melon necrotic spot virus disease in cucumbers (Tomlinson and Thomas 1986). A third species, Olpidium virulentus, can transmit olive mild mosaic virus (Reis Varanda et al. 2015). Another virus, tobacco necrosis virus, was also shown to be transmitted by Olpidium brassicae zoospores. This virus affects at least 88 different plant species, including tobacco, soybeans, and olive trees (Díaz-Cruz et al. 2017). Yikes! Fortunately for the alpine plants we study, Olpidium appears very rarely in their roots. Of the 35 plants we surveyed in 2016, only 5 of them contained Olpidium. When we do see it, the Olpidium cells are easily recognizable due to their distinct hexagonal shape (Figure 1). It remains unclear if this is an important pathogen for alpine plants or if it is causing them any harm.
References: Díaz-Cruz GA, Smith CM, Wiebe KF, Cassone BJ. 2017. First complete genome sequence of Tobacco necrosis virus Disolated from soybean and from North America. Genome Announce.5(32): e00781-17. Tomlinson JA, Faithfull EM. 1979. Effects of fungicides and surfactants on the zoospores of Olpidium brassicae. Ann. appl. Biol.93: 13-19. Tomlinson JA, Thomas BJ. 1986. Studies on melon necrotic spot virus disease of cucumber and on the control of the fungus vector (Olpidium radicale). Ann. appl. Biol.108: 71-80. Reis Varanda CM, Santos SJ, Esteves da Clara MI, do Rosário F. Félix M. 2015. Olive mild mosaic virus transmission by Olpidium virulentus. Euro. J. Plant. Path. DOI: 10.1007/s10658-015-0593-z. by Eli Gendron Dinoflagellates are an extremely morphologically and ecologically diverse group of organisms. Dinoflagellates are mainly characterized by their two dissimilar flagella (whiplike appendages used for swimming), with a transverse flagella that beats to the side of the cell and a conventional flagella that beasts posteriorly, and a cell covering called an amphiesma or cortex (Figure 1a). Many dinoflagellates, such as Karenia brevis in particular, are capable of filling multiple ecological roles as they are capable of auxotrophic heterotrophy (meaning they can utilize both organic and inorganic compounds as food), as well as also possessing chloroplasts allowing them to perform photosynthesis as well (Figure 1b) [Vargo 2009]. Karenia brevis is most well-known for its role in harmful algae blooms (HABs) and is one of the primary organisms responsible for the phenomena known as ‘red tides’ seen off the Florida coast [Ross et al. 2010]. One of the reasons HABs of Karenia brevis are so disruptive to ecosystems is due to their ability to produce brevetoxins. Brevetoxins are neurotoxins produced by dinoflagellates such as Karenia brevis, and when their populations bloom during HABs, the concentration of these toxins can reach harmful and potentially deadly levels resulting in fish kills, coral death, and even human illness [Baden 1989]. Currently, it is not currently known for certain why Karenia brevis and other dinoflagellates produce brevetoxins. One of the more recent debates revolves around the hypothesis that brevetoxins are produced in response to osmotic stress felt by these organisms [Errera & Campbell 2011; Sunda et al. 2013]. 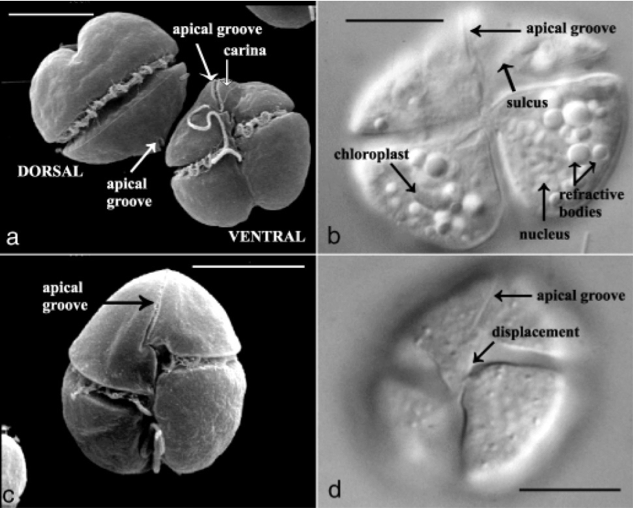 Figure 1. Karenia brevis. Scanning election micrograph (a + c) of dorsal and ventral views highlights the two flagella and characteristic apical groove. Light microscopy (b + d) of Karenia brevis ventral view highlighting organelles (particularly chloroplasts). Scale bars, 10um. Figure adapted from [Haywood et al. 2004]. References:
Baden, D. G. (1989). Brevetoxins: unique polyether dinoflagellate toxins. The FASEB journal, 3(7), 1807-1817. Errera, R. M., & Campbell, L. (2011). Osmotic stress triggers toxin production by the dinoflagellate Karenia brevis. Proceedings of the National Academy of Sciences, 108(26), 10597-10601. Haywood, A. J., Steidinger, K. A., Truby, E. W., Bergquist, P. R., Bergquist, P. L., Adamson, J., & Mackenzie, L. (2004). COMPARATIVE MORPHOLOGY AND MOLECULAR PHYLOGENETIC ANALYSIS OF THREE NEW SPECIES OF THE GENUS KARENIA (DINOPHYCEAE) FROM NEW ZEALAND 1. Journal of Phycology, 40(1), 165-179. Ross, C., Ritson-Williams, R., Pierce, R., Bullington, J. B., Henry, M., & Paul, V. J. (2010). Effects of the Florida red tide dinoflagellate, Karenia brevis, on oxidative stress and metamorphosis of larvae of the coral Porites astreoides. Harmful Algae, 9(2), 173-179. Sunda, W. G., Burleson, C., Hardison, D. R., Morey, J. S., Wang, Z., Wolny, J., ... & Van Dolah, F. M. (2013). Osmotic stress does not trigger brevetoxin production in the dinoflagellate Karenia brevis. Proceedings of the National Academy of Sciences, 110(25), 10223-10228. Vargo, G. A. (2009). A brief summary of the physiology and ecology of Karenia brevis Davis (G. Hansen and Moestrup comb. nov.) red tides on the West Florida Shelf and of hypotheses posed for their initiation, growth, maintenance, and termination. Harmful Algae, 8(4), 573-584. by Adam Solon While hiking in the mountains you may come across snow that appears red or pink. This phenomenon, commonly called “red” or “watermelon” snow, is the result of microbial organisms known as algae. Algae inhabit a variety of environments – freshwater, saltwater, soil – and are single celled organisms that, like plants, use the sun for energy (photosynthesis). A particular type of algae, the snow algae, are capable of living in snow or ice during the melt season in alpine and polar environments across the Earth (Hoham and Duval, 2000). Our lab encountered them on the high, cold, windswept slopes of the Atacama Andes (Figure 1). As members of a large group of organisms known as green algae, snow algae have been to date mostly found within the genus Chlamydomonas or genus Cholormonas. Their brilliant red color is a response to the extreme cryospheric environments in which they are found (Figure. 2). In some known species of snow algae, the compound astaxanthin, a secondary metabolite known as a carotenoid, is responsible for the characteristic red pigment (Bidigare et al., 1993). The red pigmentation serves to limit exposure to the higher radiation found in high elevation and polar environments. This has a double effect of limiting cellular damage (Hagen et al., 1993) as well as alleviating major photoinhibition which is the disruption of energy production (Thomas and Duval, 1995). So, the next time you are out hiking and see some red snow realize there are a multitudes of those little critters hard at work surviving in the cold and painting an otherwise white landscape with a little color. References:
Bidigare, R. R., Ondrusek, M. E., Kennicutt, M. C., Iturriaga, R., Harvey, H. R., Hoham, R. W., & Macko, S. A. (1993). EVIDENCE A PHOTOPROTECTIVE FOR SECONDARY CAROTENOIDS OF SNOW ALGAE 1. Journal of Phycology, 29(4), 427-434. Hagen, C. H., Braune, W., & Greulich, F. (1993). Functional aspects of secondary carotenoids in Haematococcus lacustris [Girod] Rostafinski (Volvocales) IV. Protection from photodynamic damage. Journal of Photochemistry and Photobiology B: Biology, 20(2-3), 153-160. Hoham RW, Duval B (2001) Microbial ecology of snow and fresh-water ice with emphasis on snow algae. In Jones, H. G., Pomeroy, J. W., Walker, D. A., & Hoham, R. W. (Eds.). (2001). Snow ecology: an interdisciplinary examination of snow-covered ecosystems. Cambridge University Press. Thomas, W. H., & Duval, B. (1995). Sierra Nevada, California, USA, snow algae: snow albedo changes, algal-bacterial interrelationships, and ultraviolet radiation effects. Arctic and alpine research, 27(4), 389-399. by Michelle Hollenkamp If you happen across this microbe of the month, it’s unique appearance may cause you to take a step back. Young Hydnellum peckii, commonly called the bleeding tooth fungus, oozes or “bleeds” a thick, red fluid through pores on the cap of its fruiting body. The droplets are referred to as guttules. As if the characteristic isn’t scary enough, H.peckii have 2-5mm spines, similar to teeth, on the underside of their cap. As these fungi mature, they become less descript becoming turning gray or brown in color. H.peckii belong to the Aphyllophorales order and the Hydnaceae family. They are widely dispersed in Europe and the Pacific Northwest, commonly in association with conifers and can be found among moss and pine litter. They fruit in the summer months as a solitary or fused fruiting bodies. These fungi are mycorrhiza, living in symbiosis with vascular plant roots. Despite their rather unjarring appearance, H.peckii are non-toxic. However, their taste has been described as bitter pepper deeming it inedible. This fungus does have several beneficial uses. The mushroom of H. peckii contains atromentin, which has antibacterial and anticoagulant properties. Additionally, the oozing red “blood” can be used to dye fabrics. Though these species are not found in Colorado, other Hydnellum species can be seen in the Rocky Mountains.
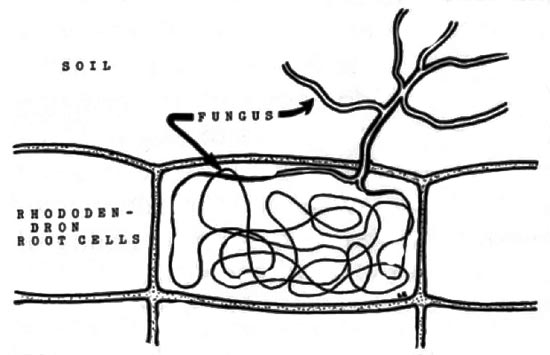
References “Bleeding Tooth Fungus.” National Geographic, 2015. <http://www.nationalgeographic.com.au/nature/the-bleeding-tooth-fungus.aspx> Evenson, Vera Stucky. “Mushrooms of Colorado and the Southern Rocky Mountains.” Big Earth Publishing, 1997. Holden, Liz. “Scotland’s rare tooth fungi: an introduction to their identification, ecology, and management. Plantlife International, 2008. by Cliff Bueno de Mesquita Leohumicola is another genus of fungi that we found frequently in the roots of the alpine cushion plant Silene acaulis (see May post). Unlike Serendipita, which I wrote about last month, Leohumicola are members of the phylum Ascomycota. Not only was Leohumicola one of the more abundant fungi in the Silene plants, it also differed among our various experimental treatments. In addition to sequencing roots of adult Silene plants from Ireland and Colorado, we conducted a greenhouse experiment in which we grew Irish Silene plants in both Irish and Colorado soil, and Colorado Silene plants in both Irish and Colorado soil. Interestingly, Leohumicola was most abundant in Colorado adult Silene plants and Irish seedling Silene plants. Leohumicola was described as a new genus in 2005, with 4 species (Hambleton et al. 2005). This group of fungi displays a remarkable range of temperature tolerances, as it has been found in high alpine environments as well as heat treated soils. One species, Leohumicola verrucosa (Figure 1) has been isolated from a pharmaceutical plant (Pleione yunnanensis) in China (Chen et al. 2009). Leohumicola have also been reported to associate with plants in the Ericaceae family, and to form ericoid mycorrhizal structures (Bizabani et al. 2016). As discussed in previous posts, mycorrhizae are fungi that associate with plants in a mutualistic relationship in which both parties benefit; the fungus receives carbon from the plant, and the plant receives nutrients from the fungus. Ericoid mycorrhizae are fungi that associate with plants in the Ericaceae family, and are characterized by forming dense coils in the plant cells for nutrient exchange (Figure 2). Researchers reported Leohumicola in commercial blueberry roots, and increased plant growth in plants with the fungus. Thus, it appears that Leohumicola could have beneficial impacts on plant growth, and potentially play a role in the growth of plants outside of the Ericaceae, like our Silene plants (family Caryophyllaceae). References:
Bizabani, C., Fontenla, S., and Dames, J.F. 2016. Ericoid fungal inoculation of blueberry under commercial production in South Africa. Scientia Horticulturae 209: 173-177. Chen, J., Meng, Z., Chen, X., Lu, Y., Zhang, F., Li, X, and Guo, S. 2009. Leohumicola, a genus new to China. Mycotaxon 108: 337-340. Hambleton, S., Nickerson, N.L., and Seifert, K.A. 2005. Leohumicola, a new genus of heat-resistant hyphomycetes. Studies in Mycology 53: 29-52. |
AuthorVarious lab members contribute to the MoM Blog Archives
October 2023
Categories |






 RSS Feed
RSS Feed
