|
by Cliff Bueno de Mesquita and Lara Vimercati Happy new year from all of us here at the Alpine (and Arctic and Antarctic) Microbial Observatory! For our first post of 2020 we will keep posting about some of the organisms in our freeze-thaw, water and nutrient amendment experiment on soils from Volcán Llullaillaco, Chile, led by Dr. Lara Vimercati. By the third water addition to microcosms receiving only water and no nutrients, we observed a significant increase in relative abundance of an OTU that was a 100% match with Neochlorosarcina negevensis, a member of the Chlorophyceae. N. negevensis is an alga commonly found in biological soil crusts of drylands (Büdel et al. 2016). Layers of algae within the Chlorophyceae have been identified as being the closest phototrophs to the soil surface within gypsum deposits at low elevations in the Atacama Desert (Wierzchos et al. 2015). Many Chlorophyceae have the capability to synthesize high amounts of complex molecules, such as carotenoids, that enhance cell resistance under unfavorable environmental conditions including excess light, UV, water and nutrition stress (Hanagata and Dubinsky, 1999; Orosa et al., 2000, 2001). Other strategies for survival in harsh conditions include the development of dormant stages or avoidance through motility (Pushkareva et al. 2016). Previous work has shown that in arid environments, high relative humidity is enough to induce metabolic activity of some algae (Palmer and Friedmann 1990); however, it is unclear whether these microalgae may be functioning in situ once water becomes available following a snowmelt event. More work will be needed to determine if Neochlorosarcina sp. is a functional phototroph in this environment. 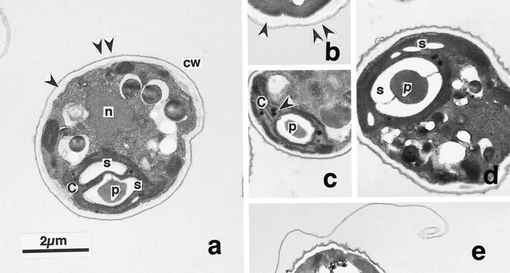 Figure 1. Transmission electron micrographs of 0-day old green cells of the alga Scenedesmus komarekii (Chlorophyceae, Chlorophyta). a. Section of vegetative cell showing distribution of major organ-elles. Arrowhead and double arrowhead show inner and outer layers of cell wall, respectively. b. Cell wall consisting of inner (arrowhead)and outer (double arrowhead) layers. c. Plastoglobuli (arrowhead) distributed in the chloroplast. d. Biplate and small starch grains inthe chloroplast. e. Curled outer layer of a parent cell wall. Abbreviations: c, chloroplast; cw, cell wall; n, nucleus; p, pyrenoid; s, starchgrain. From Hanagata and Dubinsky 1999. Of broader significance, this is the first report of phototrophs on the high elevation slopes of Volcán Llullaillaco not associated with semi-permanent water structures such as fumaroles or penitentes (Costello et al. 2009, Solon et al. 2018, Vimercati et al. 2019b). This ecosystem was previously thought to be completely heterotrophic or chemolithotrophic dependent (Lynch et al. 2014). It was believed that in this harsh ecosystem, energy entered through the system through aeolian deposited carbon sources and chemolithotrophic fixation of inorganic compounds; however, these new findings suggest that it may also enter the system via photosynthesis. However, it is important to note that this work was conducted using soils from 5100 m.a.s.l. whereas the studies mentioned above were conducted on soils from between 5300 and 6300 m.a.s.l. Therefore, more work is needed to determine if there is an elevational limit to phototrophic life not associated with penitentes or fumaroles on these volcanoes.
References: Costello, E.K., S.R.P. Halloy, S.C. Reed, P. Sowell, S.K. Schmidt. 2009. Fumarole-supported islands of biodiversity within a hyperarid, high-elevation landscape on Socompa Volcano, Puna de Atacama, Andes. Appl. Environ. Microbiol. 75: 735-747. Hanagata, N., and Dubinsky, Z., 1999. Secondary carotenoiaccumulation in Scenedesmus komarekii (Chlorophyceae, Chlorophyta). Journal of Phycology 35(5): 960-966. Lynch, R.C., Darcy, J.L., Kane, N.C., Nemergut, D.R. and Schmidt, S.K., 2014. Metagenomic evidence for metabolism of trace atmospheric gases by high-elevation desert Actinobacteria. Frontiers in microbiology, 5, p.698. Orosa, M., Torres, E., Fidalgo, P., and Abalde, J., 2000. Production and analysis of secondary carotenoids in green algae. Journal of Applied Phycology 12: 553-556. Orosa, M., Franqueira, D., Cid, A., and Abalde, J., 2001. Carotenoid accumulation in Haematococcus pluvialis in mixotrophic growth. Biotechnology Letters 23: 373-378. Palmer, R.J. and Friedmann, E.I., 1990. Water relations and photosynthesis in the cryptoendolithic microbial habitat of hot and cold deserts. Microbial ecology, 19(1), pp.111-118. Pushkareva, E., Johansen, J.R. and Elster, J., 2016. A review of the ecology, ecophysiology and biodiversity of microalgae in Arctic soil crusts. Polar Biology, 39(12), pp.2227-2240. Solon, A.J., Vimercati, L., Darcy, J.L., Arán, P., Porazinska, D., Dorador, C., Farías, M.E. and Schmidt, S.K., 2018. Microbial Communities of High-Elevation Fumaroles, Penitentes, and Dry Tephra “Soils” of the Puna de Atacama Volcanic Zone. Microbial Ecology, pp.1-12. Vimercati L., Solon, A.J., Krinsky, A., Arán, P., Porazinska, D.L., Darcy, J.L., Dorador, C., Schmidt, S.K. 2019. Nieves penitentes are a new habitat for snow algae in one of the most extreme high-elevation environments on Earth. Arct. Antarct. Alp. Res. 51(1): 190-200
0 Comments
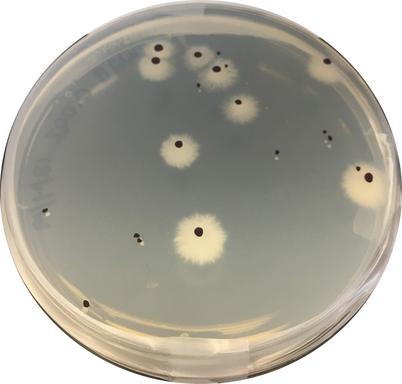
by Cliff Bueno de Mesquita Moesziomyces are one of the dominant soil fungi we found on Volcán Llullaillaco in the Atacama Desert in Chile, which is one of the most extreme environments on Earth. This high elevation site experiences high UV radiation, extreme diurnal freeze-thaw cycles, and is in the driest desert on Earth. It is possible that organisms here are dormant for most of the time but could become active during periodic snowfall events and aeolian deposition of dust containing small amounts of nutrients. In 2016, the AMO collected soils from this site and set up a microcosm experiment in a freeze-thaw chamber to assess water and nutrient limitations of the microbial community. DNA sequencing data showed that an OTU closely related to the fungal species Moesziomyces antarcticus (formerly Candida antarctica) significantly increased in relative abundance with multiple water additions to the microcosms, with the highest increase after the 3rd water addition. We just finished a follow up experiment plating these soils onto agar to grow colonies and count their abundances in the different water and nutrient additions. The plate count data corroborate the relative abundance sequencing data, showing dramatic increases in the number of colonies after 3 water additions and no nutrient additions. Species of Moesziomyces are known to produce both free-living saprobic anamorphs (yeast-like) and plant pathogenic teleomorphs (smuts) (Li et al. 2019). M. antarcticus was originally isolated from Antarctica and produces cold-active enzymes and biosurfactants (Perfumo et al. 2018). Biosurfactants have been shown to have different functions such as anti-agglomeration effects on ice particles (Perfumo et al. 2018) and are involved in cell adherence which imparts greatest stability under hostile environmental conditions. They can also be part of cellular envelopes and participate, together with other components such as exopolysaccharides, in protection against high salinity, temperature, and osmotic stress (Ewert and Deming 2013). Our study provides new evidence that along with its ability to withstand cold temperatures through cold adapted enzymes and other molecules, this yeast is also able to increase in abundance under extreme thermal fluctuations and very low nutrient contents.
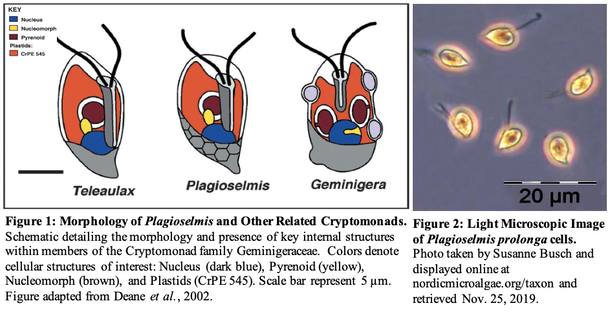
References: Ewert, M. and Deming, J., 2013. Sea ice microorganisms: Environmental constraints and extracellular responses. Biology, 2(2), pp.603-628. Li, Y., Shivas, R.G., Li, B., and Cai, L., 2019. Diversity of Moesziomyces (Ustilaginales, Ustilaginomycotina) on Echinochloa and Leersia (Poaceae). MycoKeys, 52, doi:10.3897/mycokeys.52.30461. Perfumo, A., Banat, I.M. and Marchant, R., 2018. Going green and cold: biosurfactants from low-temperature environments to biotechnology applications. Trends in biotechnology, 36(3), pp.277-289. by Eli Gendron The Cryptophyta are a group of taxonomically ambiguous dinoflagellates, which often exhibit a mixotrophic metabolism, meaning that they are often both a heterotroph (can consume organic or inorganic material for energy) and a photoautotroph (can get all their energy from light using a plastid; Figure 1; Deane et al., 2002). There is evidence that the plastid originated from the engulfment of algal cells in the phylum’s ancestry leaving behind a nucleomorph containing the ancient alga’s DNA (Figure 1; Deane et al., 2002). Cryptophytes are found in aquatic habitats around the world with the most noted freshwater family being the Geminigeraceae, with Plagioselmis being of particular interest for their potential as ecological indicators (Figure 1, 2; Markina et al., 2008). Plagioselmis species such as prolonga can be identified by their distinctive teardrop shape and twin flagella oriented on one pole of the cell (Figure 2; Olenina, 2006). Plagioselmis prolonga has been shown to respond strongly to low flow rates and increased concentrations of key nutrients such as phosphorus and nitrogen (Šupraha et al., 2014). However, Plagioselmis prolonga also is high temperature sensitive and shows a sharp decline in abundance at temperatures greater than 5oC, regardless of nutrient availability (Elliott et al., 2012). Furthermore, Plagioselmis prolonga has also shown to be sensitive to household detergents and other common detergent pollutants commonly found in freshwater and marine ecosystems (Aizdaicher et al., 2006). The sensitivity of Plagioselmis prolonga to multiple measurable environmental variables known to correlate with water quality means that including it in ecological monitoring will likely benefit future research (Markina et al., 2008). Aizdaicher, N. A., & Markina, Z. V. (2006). Toxic effects of detergents on the alga Plagioselmis prolonga (Cryptophyta). Russian Journal of Marine Biology, 32(1), 45-49.
Deane, J. A., Strachan, I. M., Saunders, G. W., Hill, D. R., & McFadden, G. I. (2002). CRYPTOMONAD EVOLUTION: NUCLEAR 18S rDNA PHYLOGENY VERSUS CELL MORPHOLOGY AND PIGMENTATION 1. Journal of Phycology, 38(6), 1236-1244. Elliott, J. A., Jones, I. D., & Thackeray, S. J. (2006). Testing the sensitivity of phytoplankton communities to changes in water temperature and nutrient load, in a temperate lake. Hydrobiologia, 559(1), 401-411. Markina, Z. V. (2008). Application of the microalga Plagioselmis prolonga for the assessment of water quality in the Amursky and Nakhodka Bays (Sea of Japan). Russian journal of marine biology, 34(1), 38-44. Olenina, I. (2006). Biovolumes and size-classes of phytoplankton in the Baltic Sea. Šupraha, L., Bosak, S., Ljubešić, Z., Mihanović, H., Olujić, G., Mikac, I., & Viličić, D. (2014). Cryptophyte bloom in a Mediterranean estuary: High abundance of Plagioselmis cf. prolonga in the Krka River estuary (eastern Adriatic Sea). Scientia marina, 78(3), 329-338. by Ylenia Vimercati Molano The genus Rhodoferax in the Comamonadaceae family comprises purple nonsulfur bacteria adapted to cold and extreme environments such as Antarctica, where the species Rhodoferax antarcticus has been isolated (Figure 1). This anoxygenic phototrophic bacterium is a psychrotrophic species (an organism capable of surviving or thriving in extremely cold environments) growing optimally at 12-18°C with a growing temperature range between 0 and 25°C (Madigan et al. 2000). 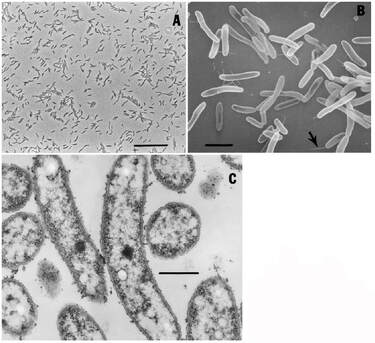 Figure 1. Morphology of cells of strain ANT.BR. (Rhodoferax antarcticus). A) Phase-contrast photomicrograph (bar 10 mm). B) Scanning electron micrograph (bar 2 mm); arrow points to possible polar flagellum. C) Transmission electron micrograph (bar 0.5 mm). Note absence of obvious intracytoplasmic membranes. From Madigan et al. 2000. Studies have reported the presence of two strains of Rhodoferax antarcticus from algal-bacterial mat samples in the Antarctica Dry Valleys as well as from limnological studies in the Vestfold Hills of East Antarctica (Jung et al. 2004). Furthermore, an ongoing project led by AMO’s Lara Vimercati at Palmer Station, Antarctica, has found a high concentration of this genus early in the chronosequence of Anvers Island’s retreating glacier. Glacial retreat in Antarctica due to climate change is a huge phenomenon and microbiologists are focused on studying the ecological succession of microorganisms along the exposed soils. According to the project, Rhodoferax is one of the most common genera in youngest, most recently deglaciated soils. The Comamonadaceae family has been observed to be dominant in stages of primary succession elsewhere too, while its abundance decreased in intermediate age soils and was absent in the oldest ones (Liu et al. 2011). We are finding this same scenario in the Palmer chronosequence, with Rhodoferax being abundant at 0 m from the glacier terminus where species richness recorded for bacteria is the lowest.
Initial stages of a glacier forefield are characterized by nitrogen and carbon deficiency (Liu et al. 2011), which makes exposed soils ideal for extreme microorganisms like Rhodoferax to colonize. As an example, Rhodoferax antarcticus can grow phototrophically (anaerobically), both photoautotrophically using hydrogen and photoheterotrophically using a variety of substrates, as well as chemoorganitrophically (aerobically in the dark) (Madigan et al. 2000). References: Jung DO, Achenbach LA, Karr EA, Takaichi S, Madigan MT. (2004). A gas vesiculate planktonic strain of the purple non-sulfur bacterium Rhodoferax antarcticus isolated from Lake Fryxell, Dry Valleys, Antarctica. Archives of Microbiol, Springer 182 :236–243 Liu G, Hu P, Zhang W, Wu X, Yang X, Chen T, Zhang M, Li S. (2011). Variations in soil culturable bacteria communities and biochemical characteristics in the Dongkemadi glacier forefield along a chronosequence. Folia Microbiol 57:485–494 Madigan MT, Jung DO, Woese CR, Achenbach LA. (2000). Rhodoferax antarcticus sp. nov., a moderately psychrophilic purple nonsulfur bacterium isolated from an Antarctic microbial mat. Archives of Microbiol, Springer 173 :269–277 by Ylenia Vimercati Mount Kilimanjaro, the tallest free-standing mountain on Earth located on the Kenya-Tanzania border, is well known for its collapsed summit forming the Reusch crater, and its permanent plateau glaciers. But, is this “island of the cryosphere” going to last long? In recent years Mt. Kilimanjaro has become an “icon” of climate change due to its dramatic ice loss that has attracted much attention among scientists, especially microbiologists, who are concerned about the fate of biological communities surviving in this harsh environment. Mt. Kilimanjaro seems to be of interest to astrobiologists too for its extreme environmental conditions considered as potential analogues for habitable zones on Mars. 85% of the ice cover has already disappeared and an increase of incoming radiation and a severe drop in atmospheric moisture are posing a threat to Mt. Kilimanjaro glaciers and their unique microbial community that may be lost forever along with the stored information on climatic and environmental changes through time. Up to now there have been no investigations published on the high-elevation microbial diversity of the habitats at the top of Mt. Kilimanjaro and on how these communities are responding to climate change drivers. This makes it extremely important and urgent to study the microbiology of the glaciers at the top of the mountain. A cutting-edge study published in July in Scientific Reports led by the AMO’s Lara Vimercati describes the diversity of soil and ice-dwelling microbes near the summit of Mt. Kilimanjaro. Field collections took place in 2012 at the border of the tabular-shaped plateau glacier in the Southern Icefield at 5772 m elevation. Surprisingly, ice and periglacial soils contain rich and diverse assemblages of Bacteria and Eukarya, probably indicating high rates of dispersal to the top of the mountain and/or that the habitat is more conducive to microbial life than previously expected. As for Bacteria, richness was high for both soil and ice communities and the high diversity encountered in the samples can be explained by glaciers supporting active microbes that gain nutrients from the atmosphere. According to the study, aeolian transportation seems crucial in the assembly of bacterial communities on Mt. Kilimanjaro. Results from the community composition analysis revealed that bacteria from soil and ice were dominated by Betaproteobacteria, especially members of the Comamonadaceae family. Scientists classified bacterial taxa into “endemic” (meaning only found on Mt. Kilimanjaro) and “non-endemic” (meaning also found elsewhere) and found that most bacterial communities within the second category were cosmopolitan. Within the Bacteria the dominant Polaromonas clade was selected to test its biogeographical distribution and analysis revealed that spatial structuring was not evident, supporting the contention that Polaromonas phylotypes are globally distributed within all glacial habitats. According to the study these Betaproteobacteria may use multiple aeolian deposited carbon sources making wind transportation fundamental for the assembly of bacterial communities. It is still unclear whether Polaromonas are indigenous to ice or they are transients from the upper atmosphere; they may be being constantly deposited in glacial systems, but not necessarily growing there.
As for Eukarya, phylogenetic analysis showed that ice communities were dominated by Cercozoa, while in soil they were the most abundant taxonomic group together with Chlorophyta. Within the Cercozoa, the authors observed that most sequences fell within the Trinematidae and the Vampirellida orders. Sequencing analysis of Vampirellida revealed that they are different from any other taxa in Genbank suggesting that this is a unique Vampirellida community with a high endemicity on Mt. Kilimanjaro probably associated with the fumarolic activity of the summit. Observations showed that they were more abundant in ice samples compared to soil samples indicating that the Vampirellida may have come from the ice. Testate amoebae abundance within the Cercozoa was high and it is probably sustained by feeding on fungi, algae and bacteria. This study is a first step towards gaining information on the global dispersal of microbes at high elevation sites in Africa. Despite the isolation of Mt. Kilimanjaro from any other mountain, the summit represents an oasis where microbes in the upper atmosphere thrive. Microbes can disperse over long distances, but they require specific adaptations to survive in high-elevation icy environments. The authors state that more work is needed to determine if these organisms can actually grow on Mt. Kilimanjaro or if they are just transient wind-dispersed microorganisms. Microbial diversity was higher than expected compared to other high volcanoes in drier regions. Bacterial community richness and diversity was quite high, and the presence of cosmopolitan bacteria suggests that the effect of distance is overwhelmed by continuous aeolian dispersal. As Mt. Kilimanjaro’s icecaps continue to vanish undisturbed, understanding the biodiversity and function of the organisms that live in the glacial and periglacial soils of the summit is extremely important and requires our attention. These ecosystems may soon disappear along with its biodiversity and pool of genes adapted to these extreme environments. The study of these life forms may also provide us a model for viable habitats for life on Mars where similar conditions may have existed or still exist. References: Vimercati L, Darcy JL, Schmidt SK (2019) The disappearing periglacial ecosystem atop Mt. Kilimanjaro supports both cosmopolitan and endemic microbial communities. Scientific Reports 9:10676 by Cliff Bueno de Mesquita and Lara Vimercati Dothideomycetes is the largest and most diverse class of fungi in the phylum Ascomycota, with 19,000 species currently described. Species within the Dothideomycetes class range from plant pathogens, to saprotrophs to some that exist as the fungal partner in the lichen symbiosis to others that live on rocks. Interestingly, we have found Dothideomycetes in some of the most extreme environments on Earth. A taxon from the Dothideomycetes class was one of the taxa that responded to our water and water + nutrient experiment in hyper-arid soils collected from ~5000 meters elevation on Volcán Llullaillaco in the Atacama Desert in Chile. In this experiment, soils in Petri dishes placed in a freeze-thaw chamber were given water or water + nutrients on three separate occasions. 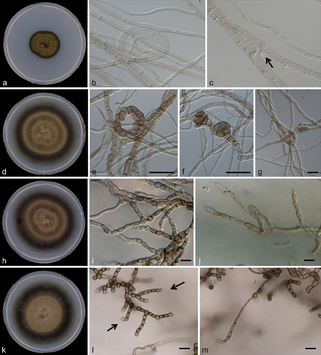 Figure 1. Dothideomycetes cultures and microscopy photos. a–c Rachicladosporium monterosium. a Colony appearance. b–c Hyphae septate, subhyaline to pale brown. d–g Rachicladosporium paucitum d Colony appearance. e–g Meristematic development of hypae, pale brown. h–j Rachicladosporium inconspicuum. From Egidi et al. 2013 Fungi within the Dothideomycete class significantly increased with multiple water additions in soil microcosms that were amended with organic carbon and nutrients. Dothideomycetes are known to be highly resistant to a number of extreme environmental conditions by developing avoidance strategies (Selbmann et al. 2015). For example, they can tolerate oligotrophy, repeated freeze-thaw stress (Onofri et al. 2007, 2008), radiation and dehydration (Onofri et al. 2012) by producing extracellular polymeric substances (EPS), melanin and compatible solutes. Many genera can form deeply melanized meristematic colonies and produce extracellular polysaccharides to ameliorate UV and osmotic stresses (Selbmann et al. 2005). Their unique colony morphology helps minimize exposure to external stressors by decreasing their surface to volume ratio and appear to have optimized a slow but persistent growth strategy for extremely cold and barren habitats (Ruibal et al. 2009). They are characterized by simple life cycles that can be completed during short periods of time when favorable conditions prevail (Selbmann et al. 2015). Oligotrophy of the extreme environments where they are found and the high metabolic costs to synthetize compounds related to stress resistance account for their slow growth velocity (Selbmann et al. 2014) and help explaining why the relative abundance of this taxon increases significantly only after the second and third water additions to the +WN treatments in this experiment, but not the initial additions.
References: Egidi, E., de Hoog, G.S., Isola, D., Onofri, S., Quaedvlieg, W., Vries, M.D., Verkley, G.J., Stielow, J.B., Zucconi, L., & Selbmann, L. (2013). Phylogeny and taxonomy of meristematic rock-inhabiting black fungi in the Dothideomycetes based on multi-locus phylogenies. Fungal Diversity, 65, 127-165. Onofri S, Barreca D, Selbmann L, Isola D, Rabbow E, Horneck G, de Vera JPP, Hatton J, Zucconi L (2008) Resistance of Antarctic black fungi and cryptoendolithic communities to simulated space and Mars conditions. Stud Mycol 61:99–109 Onofri S, de la Torre R, de Vera JP, Ott S, Zucconi L, Selbmann L, Scalzi G, Venkateswaran K, Rabbow E, Horneck G (2012) Survival of rock-colonizing organisms after 1.5 years in outer space. Astrobiology 12:508–516 Onofri S, Selbmann L, de Hoog GS, Grube M, Barreca D, Ruisi S, Zucconi L (2007) Evolution and adaptation of fungi at the boundaries of life. Adv Space Res 40:1657–1664 Ruibal C, Gueidan C, Selbmann L, Gorbushina AA, Crous PW, Groenewald JZ, Muggia L, Grube M, Isola D, Schoch CL, Staley JT, Lutzoni F, de Hoog GS (2009) Phylogeny of rock inhabiting fungi related to Dothideomycetes. Stud Mycol 64:123–133 Selbmann L, de Hoog GS, Mazzaglia A, Friedmann EI, Onofri S (2005) Fungi at the edge of life: cryptoendolithic black fungi from Antarctic deserts. Stud Mycol 51:1–32 Selbmann, L., de Hoog, G.S., Zucconi, L., Isola, D. and Onofri, S., 2014. Black yeasts in cold habitats. In Cold-adapted Yeasts (pp. 173-189). Springer, Berlin, Heidelberg. Selbmann, L., Zucconi, L., Isola, D. and Onofri, S., 2015. Rock black fungi: excellence in the extremes, from the Antarctic to space. Current genetics, 61(3), pp.335-345 by Eli Gendron The Chrysophyceae or ‘golden algae’ are a class of yellow-brown pigmented algae who get their pale coloration from the pigment fucoxanthin, which masks the darker greens of chlorophyll a + c, and generally have a motile life cycle stage where single cells develop heterokonts (pair of flagella of unequal length and often perform different functions) (Nicholls and Wujek 2003). Chrysophytes are usually found in freshwater ecosystems and associated with oligotrophic (low nutrient) environments resulting in them acting as the base nutrient source for many heterotrophic organisms (Rawson 1956). Due to their preference for oligotrophic freshwater ecosystems, members of Chrysophyceae are often abundant in the low nutrient waters of alpine lakes, streams, and rivers (Ward 1994). In particular, the Chrysophyte Hydrurus foetidusis commonly observed in glacial melt streams and other environments with low nutrients, high light availability, and rapidly moving water (Moog and Janecek 1991). H. foetidusalso shows a preference for cold water temperatures and temperatures above 16˚C are lethal (Klaveness 2017). H. foetidusis a surface attached multicellular alga (Fig.1A) which grows on stream beds and creates fibrous macro-structures (Fig.1B) which act as residence and food for invertebrate and grazing protist communities (Moog and Janecek 1991). In the attached stage of its life cycle, Hydrurusis non-motile, but when it is ready to colonize new locations, it sheds motile single cells (zoospores, Fig.1C) which possess the characteristic heterokont (Hoffman et al.1986). H. foetidus can also be found across a wide geographic range, including both Antarctica and the Arctic. Lastly, due to its wide geographic range, significant preference for oligotrophic environments, sensitivity to temperature, and its key role in nutrient availability in oligotrophic environments, H. foetidus and its relatives make ideal candidates for indicators to monitor changes in ecosystem health, stability, and trophic status under shifts in climatic conditions (Klaveness 2017).  Figure 1. Morphology of Hydrurus foetidus’ structures. Light microscope pictures of the multicellular structure (A), macrostructure (B), and illustration of the single cell zoospore (C) of Hydrurus foetidus. Scale bars are 5 µm (A), 5 mm (B), and 5 µm (C). Note the diagram of the zoospore (C) includes the heterokont flagella. The heterokont are likely an adaptation to aquatic environments as a way to facilitate the spread of offspring in order to establish new macrostructures downstream. Pictures and figures were adapted from Klaveness 2017 (A, B) and Remias et al. 2013 (C). References:
Hoffman, L.R., Vesk, M., and Pickett-Heaps, J.D. (1986) The cytology and ultrastructure of zoospores of Hydrurus foetidus (Chrysophyceae). Nord. J. Bot. 6: 105–122. Klaveness, D. (2017) Hydrurus foetidus (Chrysophyceae)—an inland macroalga with potential. J. Appl. Phycol.29: 1485–1491. Moog, O. and Janecek, B.F.U. (1991) River flow, substrate type, and Hydrurus density as major determinants of macroinvertebrate abundance, composition and distribution. Int. Vereinigung für Theor. und Angew. Limnol. Verhandlungen24: 1888–1896. Nicholls, K.H. and Wujek, D.E. (2003) Chrysophycean Algae Elsevier Inc. Rawson, D.S. (1956) Algal Indicators of Trophic Lake Types. Limnol. Oceanogr.1: 18–25. Ward, J.V. (1994) Ecology of alpine streams. Freshw. Biol.32: 277–294. by Cliff Bueno de Mesquita In our recent and ongoing experiments on the effects of herbicides on soil microbes, we found one species of fungus, Mortierella elongata, that was not negatively affected by the herbicide Glyphosate (ingredient in Roundup), and actually responded positively to the surfactant it is usually spread with, Agridex. Soils collected from the nearby Chatfield Farm outside of Denver had received treatments of Glyphosate, Agridex, Promax (another surfactant), Agridex and Glyphosate combined, and controls with no chemical additions. M. elongata is a fungus in the Mucoromycota phylum. Mortierella species are typically saprotrophs but also grow on the surface of roots. They are capable of degrading complex carbon compounds such as chitin and hemicellulose. They reproduce via formation of thick-walled spores called zygospores, which are produced by many other species of fungi as well as algae (Gams, Chien, and Domsch 2009). Typically, they are not pathogenic. 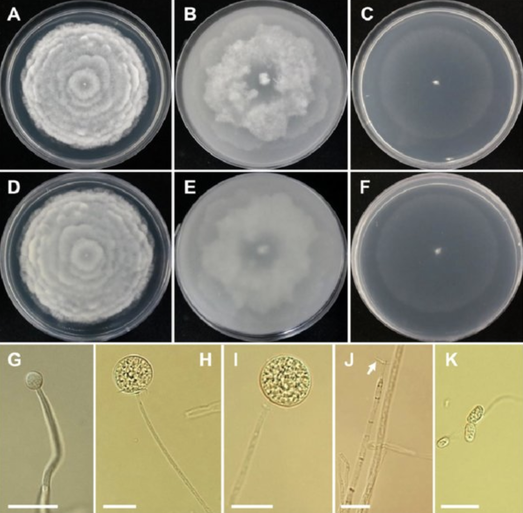 Figure 1. Morphology of Mortierella elongata CNUFC-YR329-1. A, D colony on potato dextrose agar; B, E colony on oatmeal agar; C, F colony on water agar; (A-C: observed view, D-F: reverse view). G-I young sporangia on sporangiophores; J the collar at the top of the sporangiophore (white arrow). K sporangiosphores (scale bars: G-K ¼ 20 lm). From Nguyen et al. 2019. M. elongata has been isolated and cultured. It produces Arachidonic acid (an omega-6 fatty acid also found in red meat), as well as Eicosapentaenoic acid (also known as EPA omega-3 fatty acid), both of which people take as supplements (Bajpai, Bajpai, and Ward 1991; Yamada, Shimizu, and Shinmen 1987). However, it appears that there was never large-scale commercial production of M. elongata for those purposes. Another interesting thing about this fungus is it contains a bacterium inside of it, called an endobacterium, which was identified as Mycoavidus cysteinexigens (Ohshima et al. 2016). Researchers suggested that this endobacterium was responsible for the production of nitrous oxide (Sato et al. 2010), a greenhouse gas, and there has been considerable interest in the bacterium, as its whole genome has been sequenced and assembled (Fujimura et al. 2014).
Our result with M. elongata highlights the importance of studying chemicals used to help spread herbicides, in addition to the herbicide itself. Stay tuned in future months for more results from our herbicide experiment! References: Bajpai, P., P. K. Bajpai, and O. P. Ward. 1991. “Eicosapentaenoic Acid (EPA) Formation: Comparative Studies with Mortierella Strains and Production by Mortierella Elongata.” Mycological Research 95(11): 1294–98. http://dx.doi.org/10.1016/S0953-7562(09)80577-7. Fujimura, R. et al. 2014. “Draft Genome Sequence of the Betaproteobacterial Endosymbiont Associated with the Fungus Mortierella Elongata FMR23-6.” Genome Announcements 2(6): 2010–11. Gams, W., Chiu-Yuan Chien, and K.H. Domsch. 2009. “Zygospore Formation by the Heterothallic Mortierella Elongata and a Related Homothallic Species, M. Epigama Sp.Nov.” Transactions of the British Mycological Society 58(1): 5–13. http://dx.doi.org/10.1016/S0007-1536(72)80065-2. Nguyễn, Thưởng & Won Park, Se & Pangging, Monmi & Lee, Hyang. (2019). Molecular and Morphological Confirmation of Three Undescribed Species of Mortierella from Korea. Mycobiology. 47. 31-39. 10.1080/12298093.2018.1551854. Ohshima, Shoko et al. 2016. “Mycoavidus Cysteinexigens Gen. Nov., Sp. Nov., an Endohyphal Bacterium Isolated from a Soil Isolate of the Fungus Mortierella Elongata.” International Journal of Systematic and Evolutionary Microbiology 66(5): 2052–57. Sato, Yoshinori et al. 2010. “Detection of Betaproteobacteria inside the Mycelium of the Fungus Mortierella Elongata.” Microbes and Environments 25(4): 321–24. http://joi.jlc.jst.go.jp/JST.JSTAGE/jsme2/ME10134?from=CrossRef. Yamada, Hideaki, Sakayu Shimizu, and Yoshifumi Shinmen. 1987. “Production of Arachidonic Acid by Mortierella 1S-5.” Agricultural and Biological Chemistry 51(3): 5–10. by Adam Solon When studying eukaryotic microbes, familiar groupings such as fungi, green algae, and micro animals, such as Tardigrades (water bears!), are usually found in communities. However, there also exists a large diversity of life that do not fit into those taxonomic assignments. Many are members of the broad grouping known as Protists. Protists are not united in any sort of phylogenetic connection and exist as a catchall for eukaryotic microbes that are not fungi, green algae, or animals. Among those are the organisms that make up the SAR supergroup (Stramenopiles, Alveolates, and Rhizaria). The Stramenopiles include a wide range of organisms including kelp, diatoms, and parasites such as the genus Phytophthora, infamous for causing the Great Irish Potato famine (Derelle et al. 2016). When conducting a lab experiment with soils from Antarctica we found large relative abundances of Xanthophyceae, a member of the Stramenopiles group. Xanthophyceae are a taxonomic class of the Stramenopiles and have the common name yellow-green algae. While they have the name algae, they are distantly related to the more commonly known green algae. Morphological-physiological characteristics that distinguish Xanthophyceae include: photosynthesis (i.e. use sunlight for energy, like plants), heterokonate (i.e. two flagella (threadlike appendages typically used for swimming) of different lengths, one forward tinsel flagellum and one backward whiplash flagellum); and capability of asexual or sexual reproduction (Sahoo & Kumar 2015). They also have a wide variety of characteristics in which they may differ including: uninucleate or multinucleate; unicellular, colonial or filamentous; and various combinations of the following pigments which give them a yellowish-green color- chlorophyll a and c, β-carotene and xanthophylls (Maistro et al. 2017). One of the clear distinctions between yellow-green algae and green algae is a lack of chlorophyll b in yellow-green algae.
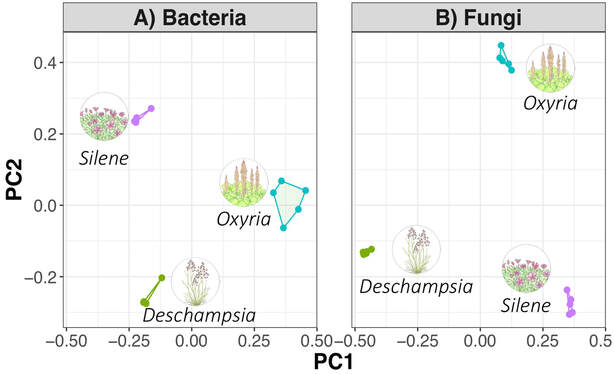
Typically found in freshwater and moist soils, with a few in marine habitats, Xanthophyceae have been discovered in both polar regions (Novis et al. 2015, Kvíderová & Elster 2017). Using soils collected from Taylor Valley in the Dry Valleys of Antarctica, we introduced inputs of water along with an initial treatment of nitrogen and phosphorus to the normally dry, oligotrophic (nutrient poor) soils from this polar desert. These little critters were able to take advantage of those nutrient additions and the subsequent water inputs and contributed to the development of a biocrust that formed on the surface. Our DNA-seq analysis revealed they made up a significant level of relative abundance in those crusts (Fig 1). References: Derelle, R., López-García, P., Timpano, H., & Moreira, D. (2016). A phylogenomic framework to study the diversity and evolution of stramenopiles (= heterokonts). Molecular biology and evolution, 33(11), 2890-2898. Kvíderová, J., & Elster, J. (2017). Photosynthetic activity of Arctic Vaucheria (Xanthophyceae) measured in microcosmos. Czech Polar Reports, 7(1), 52-61. Maistro, S., Broady, P., Andreoli, C., & Negrisolo, E. (2017). Xanthophyceae. Handbook of the Protists, 407-434. Novis, P. M., Aislabie, J., Turner, S., & McLeod, M. (2015). Chlorophyta, Xanthophyceae and Cyanobacteria in Wright Valley, Antarctica. Antarctic Science, 27(5), 439-454. Sahoo, D., & Kumar, S. (2015). Xanthophyceae, Euglenophyceae and Dinophyceae. In The algae world (pp. 259-305). Springer, Dordrecht. by Cliff Bueno de Mesquita This month I’ll write about another microbe from my litter addition experiment. One of the dominant fungi on/in the leaves of the alpine cushion plant Silene acaulis was Phaeosphaeria silenes-acaulis, which actually gets its name due to its presence on the Silene acaulis plant! Not only was this fungus abundant in Silene leaves, but it was not present in the other two plant species’ leaves that I studied, highlighting it’s specificity to Silene, although it has also been reported in a couple of other arctic/alpine plant species (Steinke and Hyde 1997, Tojo et al. 2013). The community composition of leaf fungi (and bacteria too) was significantly different in the three plant species I studied, and this was partially driven by the abundance of P. silenes-acaulis in Silene acaulis and not the other two species (Figure 1). Statistical analysis showed that P. silenes-acaulis contributed 14% to differences in fungal communities between Silene and the bunchgrass Deschampsia, and 31% to differences in fungal communities between Silene and the edible alpine mountain sorrel plant Oxyria. The genus Phaeosphaeria is in the phylum Ascomycota and contains saprotrophic fungi that function as decomposers. Sixteen different species in the genus have been previously reported in arctic and alpine ecosystems, including Svalbard, Norway and the Tatra Mountains in Slovakia (Tojo et al. 2013, Kozłowska et al. 2016). These fungi are adapted to these ecosystems by having a simple life cycle, forming thick and deeply pigmented walls, and having ascospores (reproductive spores produced by fungi in the Ascomycota phylum, Figure 2) with gelatinous sheaths (Leuchtmann 1987). Another species of note in the genus is Phaeosphaeria oryza, which associates with rice and is an important biological control agent of rice blast disease (Ohtake et al. 2008). As with many microbial taxa, we have now done the basic ecological work of sequencing and phylogenetic classification, but information is lacking on function. As I have done with some of my root samples, future work could attempt to isolate this fungus by placing Silene acaulis leaves on petri dishes with agar and an energy source, and then do further experiments to examine the role of the fungus either in decomposition or Silene acaulis plant health.
References: Bueno de Mesquita C.P., Schmidt S.K., Suding K.N. (2019) Species-specific plant-microbe interactions in an early successional ecosystem. Plant and Soil in review. Kozłowska M., Mułenko W., Bacigálová K, Wołczańska A., Świderska-Burek U., Pluta M. (2016) Microfungi of the Tatra Mountains. Part 7. Correction of some data from herbaria and the literature. Acta Mycologica 51(2):1081. Leuchtmann A. (1987) Phaeosphaeria in the Arctic and Alpine Zones. In: Laursen G.A., Ammirati J.F., Redhead S.A. (eds) Arctic and Alpine Mycology II. Environmental Science Research, vol 34. Springer, Boston, MA Ohtaka N., Kawamata H., Narisawa K. (2008) Suppression of rice blast using freeze-killed mycelia of biocontrol fungal candidate MKP5111B. Journal of General Plant Pathology 74: 101-108 Quaedvlieg W., Verkley G.J., Shin H.D., Barreto R.W., Alfenas A.C., Swart W.J., Groenewald J.Z., Crous P.W. (2013) Sizing up Septoria. Studies in Mycology 75: 307-390. Tojo M., Masumoto S., Hoshino T. (2013) Phytopathogenic Fungi and Fungal-Like Microbes in Svalbard. In: Imai R., Yoshida M., Matsumoto N. (eds) Plant and Microbe Adaptations to Cold in a Changing World. Springer, New York, NY Steinke T.D., Hyde K.D. (1997) Phaeosphaeria capensis sp. nov. from Avicennia marina in South Africa. Mycoscience 38: 101-103. |
AuthorVarious lab members contribute to the MoM Blog Archives
October 2023
Categories |



 RSS Feed
RSS Feed
