|
by Cliff Bueno de Mesquita Serendipita is a genus of mycorrhizal fungi from a different lineage than most other mycorrhizae. Remember, a mycorrhizal fungus is one that forms a mutualism with a plant, helping the plant uptake nutrients in exchange for carbon that the plant creates through photosynthesis. Serendipita are in the order Sebacinales, class Agaricomycetes, phylum Basidiomycota. Fungi in the order Sebacinales were originally considered “orchid mycorrhizae”, as they were thought to only associate with orchid plants. However, they have now been found in mosses, ferns, and a variety of other plants including wheat, maize, poplar, and tomato. In our own work, we found that Serendipita was a dominant fungus found inside the roots of the moss campion Silene acaulis, a beautiful arctic-alpine plant with a circumpolar distribution (Figure 1A, 1B). We sequenced the fungal DNA found inside the roots of Silene acaulis from plants at our site in Colorado, as well as from plants from Ireland, in collaboration with a visiting Fulbright scholar Conor Meade, from the University of Maynooth, Ireland. We defined “dominant” as a fungus being in the top 10% most abundant fungi in the roots, and found in over 50% of the individual plants sampled (Delgado-Baquerizo et al. 2018). Serendipita was one of only four known fungal genera that qualified as dominant, so we think it is playing a role in the growth of Silene acaulis. The genus Serendipita was first described in 1993 (Roberts 1993). One of the species in the genus, Serendipita vermifera, has been shown to increase the growth of switchgrass, which is grown as a bioenergy crop (Ray et al. 2018). Another species, Serendipita indica, was shown to increase the growth of the plant used to make turmeric (Bhola et al. 2017). Researchers are currently working on growing this fungus in large quantities to then add to soil to promote plant growth (Singhal et al. 2017). References: Bhola, D., Bajaj, R., Tripathi, S., Varma, A. 2017. Piriformospora indica(Serendipita indica) enhances growth and secondary metabolites in Cucurma longa. In: Varma, A., Prasad, R., Tuteja, N., (eds). Mycorrhiza – Nutrient Uptake, Biocontrol, Ecorestoration. Springer, Cham. Delgado-Baquerizo, M., Oliverio, A.M., Brewer, T.E., Benavent-González, A., Eldridge, D.J., Bardgett, R.D., Maestre, F.T., Singh, B.K., Fierer, N. 2018. A global atlas of the dominant bacteria found in soil. Science359: 320-325. Ray, P., Guo, Y., Sun, L., Tang, Y., Craven, K.D., Gilna, P. 2018. Signaling between switchgrass and fungal endosymbionts in the genus Serendipita. https://genomicscience.energy.gov/pubs/2017abstracts/abstractpdfs/73_Gilna_Paul_Ray.pdf Roberts P. 1993. Exidiopsis species from Devon, including the new segregate genera Ceratosebacina, Endoperplexa, Microsebacina, and Serendipita. Mycological Research. 97 (4): 467–78. doi:10.1016/S0953-7562(09)80135-4. Singhal, U., Attri, M.K., Varma, A. 2017. Mass cultivation of mycorrhiza-like fungus Piriformospora indica(Serendipita indica) by batch in bioreactor. In: Varma, A., Prasad, R., Tuteja, N. (eds). Mycorrhiza – Function, Diversity, State of the Art. Springer, Cham. Vohník, M., Pánek, M., Fehrer, J., Selosse, M. 2016. Experimental evidence of ericoid mycorrhizal potential within Serendipitaceae (Sebacinales). Mycorrhiza26(8): 831-846.
0 Comments
by Eli Gendron One of the greatest challenges sessile (immobile) organisms face is obtaining an adequate supply of food while not being able to actively forage or hunt for it. One way to get around this limitation is to have a motile life stage that is able to actively disperse to other parts of the environment where food might be more abundant (Sousa 1979). Members of the Vorticella genus are an example of ciliates that possess this adaptation, and thrive in a wide variety of freshwater habitats, and even inundated grasslands (Sun et al. 2012; Stout 1984). Ciliates are single-celled organisms that make up their own phylum (Ciliophora), and are characterized by having cilia, or hair-like structures on the outside of their cells. Vorticella species alternate between a free-swimming motile stage known as a telotroch, and a sessile phase known as a trophont, where it forms a contractible stalk that anchors the body to a substrate (Williams and Clamp 2007). The contractible stalk is one example how this genus has adapted to freshwater ecosystems. The stalk has been hypothesized to allow the body to retract closer to the substrate during turbulent conditions or potentially to mix its surroundings in order to obtain food in extremely calm conditions (Ryu et al. 2016, Figure1C). Vorticella sp. are very effective filter feeders (feeding by straining substances from the water) due to their ability to use their cilia to create currents that direct nearby organic matter directly into their body cavity (Ryu et al. 2016, Figure 1B). Taken together, these adaptations have made Vorticella sp. a target for bioengineering microfluidic devices. Outside of engineering applications Vorticella sp. even have potential applications as biocontrol agents for mosquitos. Patil et al. 2016 found that mosquito larvae that became infected with parasitic a Vorticella sp. saw a 90% reduction in adult emergence. Lastly, Vorticella’s ubiquity in freshwater habitats and their sensitivity to water quality make them excellent candidates for indicators of ecosystem health. For example, Vorticella sp. have been observed thriving in aquatic habitats with high levels of organic matter and low nitrogen cycling capacity (Perez-Uz et al. 2010).
References: Patil CD, Narkhede CP, Suryawanshi RK, Patil SV. 2016. Vorticella sp: Prospective mosquito biocontrol agent. J Arthropod Borne Dis 10:602–607. Pérez-Uz B, Arregui L, Calvo P, Salvadó H, Fernández N, Rodríguez E, Zornoza A, Serrano S. 2010. Assessment of plausible bioindicators for plant performance in advanced wastewater treatment systems. Water Res 44:5059–5069. Ryu S, Pepper RE, Nagai M, France DC. 2017. Vorticella: A protozoan for bio-inspired engineering. Micromachines 8:1–25. Stout JD. 1984. The protozoan fauna of a seasonally inundated soil under grassland. Soil Biol Biochem 16:121–125. Sun P, Clamp J, Xu D, Kusuoka Y, Miao W. 2012. Vorticella Linnaeus, 1767 (Ciliophora, Oligohymenophora, Peritrichia) is a Grade not a Clade: Redefinition of Vorticella and the Families Vorticellidae and Astylozoidae using Molecular Characters Derived from the Gene Coding for Small Subunit Ribosomal RN. Protist 163:129–142. Williams D, Clamp JC. 2007. A molecular phylogenetic investigation of Opisthonecta and related genera (Ciliophora, Peritrichia, Sessilida). J Eukaryot Microbiol 54:317–323. by Lara Vimercati Extremophilic (organisms that live in extreme environments) members of the yeast Naganishia (previously classified as the Cryptococcus genus) represent the most abundant eukaryotes (organisms with a nucleus) from some of the harshest ecosystems yet discovered on Earth: the high elevations in dry valleys and slopes of the high Andes (Costello et al. 2009, Lynch et al. 2012, Vimercati et al. 2016). This Naganishia sp. (Phylum Basidiomycota) is most closely related to Cryptococcus friedmannii, which lives on rocks and tolerates extreme droughts, from the Dry Valleys of Antarctica and is also related to some of the dominant microeukaryotes of other extreme polar and glacial regions (Buzzini et al. 2012). Recent studies indicate that Naganishia species from the high Andes are among the most resistant organisms to UV radiation (Pulschen et al. 2015) and our work on a Naganishia strain from the high Andes showed that it is able to grow continuously in liquid culture when exposed to extreme thermal fluctuations. In this experiment we simulated the diurnal freeze-thaw cycles that occur on a continuous basis at elevations above 6000 meters above sea level in the Atacama region cycling up to a high of 30°C during the day and down to a low of -10°C at night, an amplitude of 40°C in a 24-hour period (Vimercati et al. 2016). These studies provide evidence that Naganishia sp. has the capacity to grow during freeze-thaw cycles in the field, and that it may be able to mainly do that during periods of higher soil moisture. Given its unique ability to survive under multiple environmental stressors, Naganishias may be a model organism for astrobiology and studies on stress resistance in eukaryotes, and its adaptive strategies could be crucial as predictive tools in investigating the limits of life. 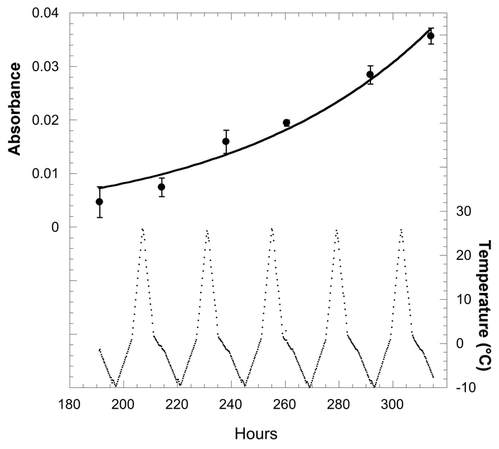 Figure 1. Exponential growth of the Naganishia sp. isolate (top panel) during freeze-thaw cycles of 27°C to -10°C as measured with data loggers in the growth chamber (bottom panel). Higher absorbance values mean more growth. Temperatures in the actual growth medium were comparable to these in the chamber, but showed a lag of about 1 hour (data not shown). All cultures froze solid every night and completely melted every day. It is not clear from these data if growth was continuous or occurred only during periods of liquid water since all sub-samples were taken when cultures were melted. Each point represents the mean of four replicate cultures (error bars are standard error of the mean). References:
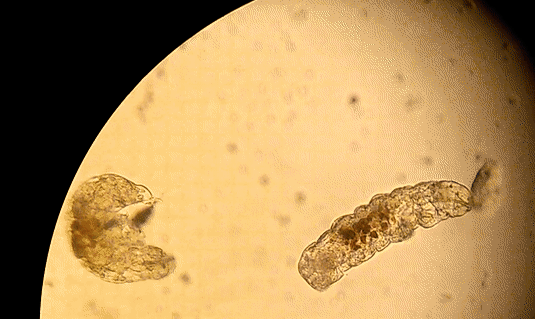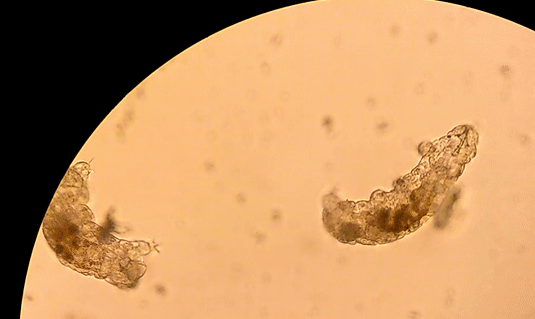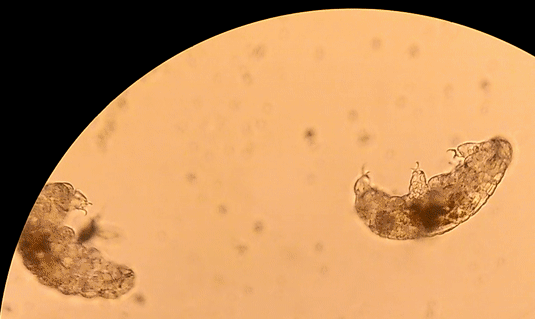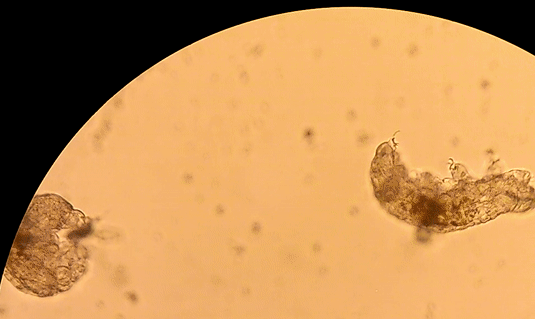
Buzzini P, Branda E, Goretti M, Turchetti B (2012) Psychrophilic yeasts from worldwide glacial habitats: diversity, adaptation strategies and biotechnological potential. FEMS Microbiol Ecol 82: 217–241. Costello, E.K., S.R.P. Halloy, S.C. Reed, P. Sowell, S.K. Schmidt. 2009. Fumarole supported islands of biodiversity within a hyperarid, high-elevation landscape on Socompa Volcano, Puna de Atacama, Andes. Appl. Environ. Microbiol. 75: 735-747. Lynch, R.C., King, A.J., Farías, M.E., Sowell, P., Vitry, C. and Schmidt, S.K., 2012. The potential for microbial life in the highest‐elevation (> 6000 masl) mineral soils of the Atacama region. Journal of Geophysical Research: Biogeosciences, 117(G2). Pulschen, A.A., Rodrigues, F., Duarte, R.T., Araujo, G.G., Santiago, I.F., Paulino‐Lima, I.G., Rosa, C.A., Kato, M.J., Pellizari, V.H. and Galante, D., 2015. UV‐resistant yeasts isolated from a high‐altitude volcanic area on the Atacama Desert as eukaryotic models for astrobiology. MicrobiologyOpen, 4(4), pp.574-588. Vimercati L, Hamsher S, Schubert Z, Schmidt SK. 2016. Growth of a high-elevation Cryptococcus sp. during extreme freeze-thaw cycles. Extremophiles. 20:579–588. by Pacifica Sommers Tardigrades, also known as “water bears” or “moss piglets,” are microscopic animals that have captured the public’s imagination in such forums as the new Star Trek: Discovery television series (https://www.space.com/38477-star-trek-discovery-mudd-ies-up-tardigrade-science.html) for their ability to survive the radiation of space (Rebecchi et al. 2009). Although most microbiologists would consider them microfauna, or microscopic animal life, rather than microbes strictly speaking, we are including them here because they are invisible to the naked eye. You typically need a microscope to see these creatures less than one millimeter long. However, like humans rather than bacteria, tardigrades have nuclei in their cells, and are even multicellular – that is, they are made up of many cells (Nichols 2005). Tardigrades have a piercing mouth part that some herbivorous species use to eat mosses or plant-like algae, and other predatory species use to eat microscopic animals such as nematodes or rotifers (Nichols 2005, Hohberg & Traunspurger 2009). When placed on a glass or plastic petri dish or slide to view under a microscope, tardigrades have a hard time gaining traction, and thus look like they are running in place, moving slowly: hence the name tardigrade, which means “slow stepper.” However, if they have sand or something sticky in a petri dish like agar, they can walk somewhat like an eight-legged caterpillar might. They live in many environments around the world, especially in mosses, in leaf litter, in the nutrient-poor soils of polar and alpine environments, and can even be found in the ocean (Nichols 2005). The tardigrades pictured here were extracted from cryoconite holes, or small mud puddles, on Antarctic glaciers. References
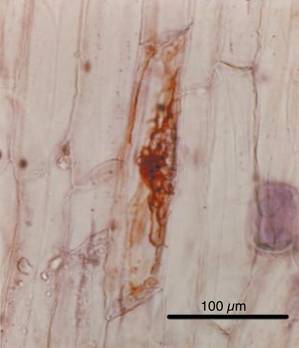
Hohberg, K., & Traunspurger, W. (2009). Foraging theory and partial consumption in a tardigrade– nematode system. Behavioral Ecology, 20(4), 884-890. Nichols, P. B. (2005). Tardigrade evolution and ecology. Dissertation, University of South Florida. Rebecchi, L., Altiero, T., Guidetti, R., Cesari, M., Bertolani, R., Negroni, M., & Rizzo, A. M. (2009). Tardigrade resistance to space effects: first results of experiments on the LIFE-TARSE mission on FOTON-M3 (September 2007). Astrobiology, 9(6), 581-591. by Cliff Bueno de Mesquita Phialophora is a genus of fungus containing taxa capable of a variety of functions. They can be parasitic on fruits and humans, saprotrophic (decompose dead plant material), or mutualistic with plants. At our field site at Niwot Ridge in the Colorado Rocky Mountains, Phialophora was one of the most abundant fungi we found living inside of plant roots. We found DNA sequences of this genus in 126 of 177 plants that we sampled. Before extracting the DNA from plant roots, we washed them in ethanol and bleach to sterilize the root surface; thus we believe this fungus is inhabiting the inside of plant roots and directly interacting with the plant. We are interested in this fungus because it is a dark septate fungus that can benefit plant growth. For example, Newsham (1999) found that the fungus Phialophora graminicola benefited the growth of its host plant. We’ve known that this group of fungi lives in the alpine for a while now. A former student in the lab, Chris Schadt, found a fungus in this genus (Phialophora gregata) inside of an alpine plant back in 2001 (Fig. 1). Interestingly, the sequences from our 2016 survey at Niwot were closely related to Phialophora that were found inside of moss tissues on King George Island in Antarctica, which suggests that this genus is widespread geographically. We are currently trying to culture this fungus to isolate it and conduct experiments with it in the greenhouse to see if this particular strain benefits plant growth.
References Newsham, K. K. 1999: Phialophora graminicola , a dark septate fungus, is a beneficial associate of the grass Vulpia ciliata ssp. ambigua. New Phytologist, 144: 517–524, doi: https://doi.org/10.1046/j.1469-8137.1999.00537.x. Newsham, K. K. 2011: A meta-analysis of plant responses to dark septate root endophytes. New Phytologist, 190: 783–793, doi: https://doi.org/10.1111/j.1469-8137.2010.03611.x. Schadt, C. W., Mullen, R. B., and Schmidt, S. K. 2001: Isolation and phylogenetic identification of a dark-septate fungus associated with the alpine plant Ranunculus adoneus. New Phytologist, 150: 747–755. Yu, N. H., Kim, J. A., Jeong, M.-H., Cheong, Y. H., Hong, S. G., Jung, J. S., … Hur, J.-S. 2014: Diversity of endophytic fungi associated with bryophyte in the maritime Antarctic (King George Island). Polar Biology, 37: 27–36, doi: https://doi.org/10.1007/s00300-013-1406-5. Mortierella spp. by Dorota Porazinska Mortierella species are aseptate, filamentous soil fungi belonging to the order of Mortierellales of the Zygomycota phylum. Among the 70+ described species, most are commonly found in soil. As saprophytic organisms (those that live on dead or decomposing matter), they can be associated with a multitude of substrate sources including plant litter. A unique feature of Mortierella is that some species are known to be exceptionally well adapted to living under very cold conditions typical of arctic, Antarctic and high alpine snow-covered soils. As psychrophilic (cold-loving), they can grow at near- or even below-freezing temperatures and survive freeze-thaws cycles and desiccation. To tolerate cold, they employ physiological mechanisms that diminish the possibility of cellular water to freeze, crystalize, and rupture cell membranes. The most common mechanisms include production of trehalose and cryoprotectant sugars that stabilize membranes, glycerol and mannitol that maintain turgor pressure, unsaturated fatty acids that maintain fluidity of membrane structures, and antifreeze proteins that may slow the growth of ice crystals. At our field site at Niwot Ridge in the Colorado Rocky Mountains, Mortierella species can be easily observed as white mats, also known as “snow molds”, that quickly appear at the edges of receding snow banks in the spring time. Their incredible fast growth rates have been attributed to their ability to rapidly exploit a fresh flush of nutrients stored in soil and water from melting snow. However, as soon snow is gone, they disappear as fast as they appeared. In a recent survey of soil communities from a range of soil habitats spanning bare to increasingly vegetated, the abundance of Mortierella spp. declined where plant communities have become more established. Because increasing incidence of plants in this landscape reflects diminishing snow cover likely due to climate warming, the future of cold-loving Mortierella spp. lies in “hands” of surviving snow packs keeping high alpine ecosystems free of vegetation.
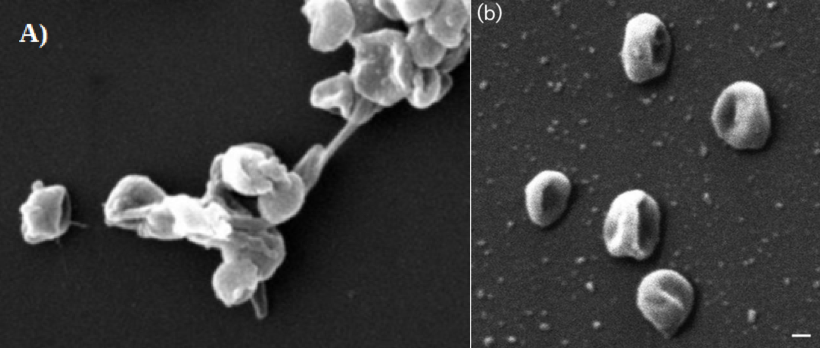
Literature Resources Schmidt SK, Wilson KL, Meyer AF, Gebauer MM, King AJ. 2008. Phylogeny of ecophysiology of opportunistic “snow molds” from a subalpine forest ecosystem. Microbial Ecology, 56: 681-687. Robinson CH. 2001. Cold adaptation in Arctic and Antarctic fungi. New Phytologist, 151: 341-353. Nitrososphaera viennensis By Eli Gendron The microbe we will be covering this month comes to us from our nearest neighboring domain on the tree of life: The Archaea. While many think of the Archaea rare in today’s world, the truth is that Archaeal species can be found in almost every environment we look. Nitrososphaera viennensis can even be found in everyday soils that are located in our own backyards, and was originally isolated from garden soil in Vienna, Austria (Stieglmeier et al. 2014). While displaying unique morphological traits (Figure 1) N. viennensis is interesting to researches and the public alike for its ability to feed on ammonium (NH4+). Research has already shown that a close relative of N. viennensis known as Nitrososphaera gargensis can be a potential tool in combating nitrogen pollution in water[1]. N. viennensis has even been found in manure piles produced from farmland waste[5] and may be important in the breakdown of ammonium in fertilizers. I would like to highlight that due to the worldwide distribution of these organisms, and their relatives, they play a key role in the global nitrogen cycle. Nitrogen pollution produced by human activity has severally altered the world’s nitrogen cycle[2]. Thus understanding how these organisms’ ecology will help us understand how we have impacted the natural world and how we might be able to look back to that world to find solutions.
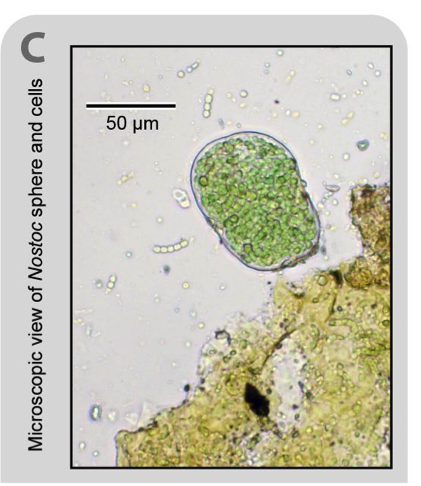
References: 1. Courtens, E. N., Spieck, E., Vilchez-Vargas, R., Bodé, S., Boeckx, P., Schouten, S., … Boon, N. (2016). A robust nitrifying community in a bioreactor at 50 °c opens up the path for thermophilic nitrogen removal. ISME Journal, 10(9), 2293–2303. http://doi.org/10.1038/ismej.2016.8 2. Galloway, J. N., Leach, A. M., Bleeker, A., & Erisman, J. W. (2013). A chronology of human understanding of the nitrogen cycle. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 368(1621), 20130120. http://doi.org/10.1098/rstb.2013.0120 3. Stieglmeier, M., Klingl, A., Alves, R. J. E., Rittmann, S. K. M. R., Melcher, M., Leisch, N., & Schleper, C. (2014). Nitrososphaera viennensis gen. nov., sp. nov., an aerobic and mesophilic, ammonia-oxidizing archaeon from soil and a member of the archaeal phylum Thaumarchaeota. Inter. J. of Sys. and Evo. Micro., 64(8), 2738–2752 4. University of Vienna. (2011, April 28). Novel microorganism 'Nitrososphaera viennensis' isolated.ScienceDaily. Retrieved November 29, 2017 from www.sciencedaily.com/releases/2011/04/110425153556.htm 5. Yamamoto, N., Oishi, R., Suyama, Y., Tada, C., & Nakai, Y. (2012). Ammonia-Oxidizing Bacteria Rather than Ammonia-Oxidizing Archaea were Widely Distributed in Animal Manure Composts from Field-Scale Facilities. Microbes and Environments, 27(4), 519–524. http://doi.org/10.1264/jsme2.ME12053 Nostoc commune var sphaeoroides by Jack Darcy Every living thing needs it, but nitrogen (N) is tough to get in nature. On earth our primary source of N is the air, which is 80% N. But atmospheric N is very difficult for living things to use, because it consists of two N atoms triple-bonded together. Getting them apart, so they can be bonded to other atoms, requires a LOT of energy. Fortunately, Cyanobacteria of the genus Nostoc are famous worldwide for their ability to convert atmospheric N into a biologically useful form. Although other microbes can do this “N-fixing” too, these photosynthetic bacteria are special because they fix N, they fix C (they turn atmospheric carbon dioxide into their food!), and they are really cool-looking. Some of these Nostoc grow as spherical colonies (see picture), which are eaten by people in Peru and Chile. We call these strains Nostoc commune var. sphaeroides, but in Peru they are called “algas de la laguna”, “cushuro”, and “llullucho”. The edible forms of these cyanobacteria are commonly found in streams, but their close relatives have been found in much more extreme environments. In our expeditions to the Peruvian Andes, we’ve found tiny (less than 1mm in diameter) Nostoc spheres in dry, plant-free soil that was recently uncovered by a retreating glacier (Darcy et al. 2018). We also found related Nostoc species at extremely high-elevation, above 5500 meters above sea level (18,100 feet), where the air is very cold, it’s very dry, and only the toughest microbes can survive (Schmidt et al. 2017). Aspergillus spp. By Cliff Bueno de Mesquita September’s microbe of the month is the genus of fungi Aspergillus. Aspergillus is a mold type of fungus and is another fungus that we’ve found in the roots of alpine plants on Niwot Ridge, Colorado. However, the genus is very widespread, and can be found indoors as well. Interestingly, it was also one of the few fungal taxa that we found in air samples that we collected at Niwot Ridge last summer. We put up air filters (Figure 1) that trap fungal spores and other microbes to examine potential dispersal limitation of microbes. In other words, are microbes able to be blown all over the landscape, or are they only able to reach some random locations? Unlike some of the other fungi I have written about, Aspergillus is typically pathogenic to plants and can even cause disease in humans and other animals too. While mycorrhizae take carbon from plants, they give the plants nutrients in return and have a beneficial effect. Pathogens like Aspergillus take resources from plants without giving them anything in return. One of the most abundant species in the genus, Aspergillus fumigatus, can cause pulmonary infections in humans. There has been a lot of work on Aspergillus in the agricultural field, as some species have been known to cause disease on important crops such as corn. References: CDC. 2015. Sources of Aspergillosis. https://www.cdc.gov/fungal/diseases/aspergillosis/causes.html Pioneer. 2017. Aspergillus ear rot. https://www.pioneer.com/home/site/us/agronomy/crop-management/corn-insect-disease/aspergillus-ear-rot/ The Aspergillus website. http://www.aspergillus.org.uk/ Amanita muscaria by Cliff Bueno de Mesquita In keeping with the summer’s fungus theme, this month’s microbe is Amanita muscaria, another one that we see commonly in the Colorado subalpine on our way up to sites at the Niwot Ridge Long Term Ecological Research site. Amanita is straight out of the Super Mario games or Alice in Wonderland – the classic red mushroom with white spots. Unlike the King Bolete I wrote about last month, I do not recommend eating the Amanita, as it is highly toxic, containing ibotenic acid and muscimol compounds. In contrast to the spongy King Bolete, the Amanita has gills on the underside of the cap, which is the first sign that you should be wary of eating it. Amanita muscaria is one of 600 species in the Amanita genus. They are in the Amanitaceae family, in the Agaricales order (gilled mushrooms) in the Basidiomycota phylum. Amanita muscaria is mycorrhizal, and associates with both hardwoods and conifers. In Colorado, it appears to be associating with the subalpine fir, all the way up to treeline, and produces mushrooms in July and August after it rains. It ranges from Mexico all the way to Alaska. The cap is 5-25 cm in diameter, deep to bright red, with yellow “warts” that quickly fad to white.
References: http://www.mushroomexpert.com/amanita_muscaria_flavivolvata.html http://www.mykoweb.com/CAF/species/Amanita_muscaria.html |
AuthorVarious lab members contribute to the MoM Blog Archives
October 2023
Categories |







 RSS Feed
RSS Feed
